Energy Efficient Housing Project:
This Energy Efficient Housing has been our longest and biggest project of the year. We accomplished 6 "mini" projects, while also learning about some major topics along the way. We were assigned to create a Solar Water Heater, where we had to have the most temperature increase in the water. We did a daylighting design project where we had to get the most possible light into a 1,000 square foot cardboard house that we designed. We studied important elements that go into site selection when building houses. As a class we did a materials testing lab, in which we figured out which materials were conductive and which materials weren't. In our individual groups we had to design a cold frame and a solution to our North Window. For the justification project we researched alternative forms of energy as a class on a shared google document. Throughout all of these projects we also learned about sun angles, and atomic structure, state, and heat.
SOLAR WATER HEATER:
For our project we had to maximize the material that was given to us to have the most heat increase in our water. We received 3 feet of a copper pipe, and had to bring the rest of our materials from home. My group brought a water bottle, saran wrap, a rag, and aluminum foil. We got a box out of the school recycling as well. We then went on to design our water heater. We painted the copper pipe black so that it would attract more sunlight (heat). We also put a lining of aluminum foil along the inside of our box to reflect the sunlight onto the copper tubing from multiple different angles. We also added a reflector to maximize the given sunlight. We added saran wrap to add a "greenhouse effect" which would trap the heat.
Our design worked fairly well. The copper pipe conducted the heat, making the water warm up. We definitely felt an increase of heat. The major flaw in our design was that our pouring/collecting water was very sloppy, resulting in a tremendous loss of water in the heating process. That being said, our box was angled at 64 degrees, allowing the sunlight to come in without shading the box too much. We also added a reflector with three flaps around it to reflect as much light as possible onto our copper piping. I thought that our group worked well together, and that overall, our project was pretty strong.
We also learned about heat transfer.
Conduction: transfer of heat through solids
Convection: transfer of heat through fluids
Radiation: transfer of heat through waves/particles/rays
Our design worked fairly well. The copper pipe conducted the heat, making the water warm up. We definitely felt an increase of heat. The major flaw in our design was that our pouring/collecting water was very sloppy, resulting in a tremendous loss of water in the heating process. That being said, our box was angled at 64 degrees, allowing the sunlight to come in without shading the box too much. We also added a reflector with three flaps around it to reflect as much light as possible onto our copper piping. I thought that our group worked well together, and that overall, our project was pretty strong.
We also learned about heat transfer.
Conduction: transfer of heat through solids
Convection: transfer of heat through fluids
Radiation: transfer of heat through waves/particles/rays
ATOMIC STRUCTURE, STATE, AND HEAT
To begin to design our solar water heater, we had to learn about a few major concepts first. We studied the atomic structure, state, and heat. We also learned about radiation, convection, and conduction. The 4 main components are the nucleus, protons, neutrons, and electrons. Protons have a positive charge, electrons have a negative charge, and neutrons are neutral. We learned that molecules have two or more atoms working together. In one cubic centimeter of air there is 45 billion billion molecules. We also learned about heat. A cold water molecule, for example is dense, and the atoms inside of it are moving slowly. A hot water molecule, for example is less dense, and the atoms inside of it are moving quickly. Evaporation is the cooling process for water. The water vapor molecules that evaporate take kinetic energy/heat with them, leaving less heat with the remaining water.
Heat is the measurement of the movement of molecules. (more heat=more movement) All molecules are held in place if they are a solid. All molecules are bonding with each other but they can change bonds to flow if they are in a liquid state. If it's a gas, molecules don't have to/can't bond with each other, which means more movement, and takes up way more space. If it's in a gas state it's also less dense.
We also learned about heat transfer.
Conduction: transfer of heat through solids
Convection: transfer of heat through fluids
Radiation: transfer of heat through waves/particles/rays
Heat is the measurement of the movement of molecules. (more heat=more movement) All molecules are held in place if they are a solid. All molecules are bonding with each other but they can change bonds to flow if they are in a liquid state. If it's a gas, molecules don't have to/can't bond with each other, which means more movement, and takes up way more space. If it's in a gas state it's also less dense.
We also learned about heat transfer.
Conduction: transfer of heat through solids
Convection: transfer of heat through fluids
Radiation: transfer of heat through waves/particles/rays
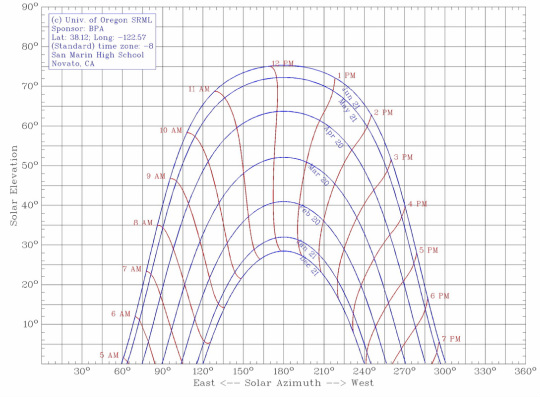
SUn Angles
|
We were given a graph of the sun's angles/ positions throughout different times of the year. Using the knowledge that we gained, we were able to correctly angle our solar water heater to the sun to get the most possible light onto our copper pipe. (This knowledge helped us with the daylighting design and techniques as well. )
|
Daylighting design and techniques:
We learned about alternative forms of lighting that require much less energy, compared to the usual light bulb. We learned about solar tubes, skylights, light shelves, and clerestory windows. Our first assignment was to calculate the amount of kilowatts we use in our homes now, and then replace some lights with solar tubes, skylights, and celestory windows. For example, I use about 7.13 kilowatts now, and if I were to use alternative forms of lighting, I would use about 3.43 kilowatts. The equation we used to calculate the kilowatts was:
(# of bulbs) × (Bulb wattage) × (average # of hours per bulb)
(1000 watts/kilowatt )
As a group we had to design a 1,000 square foot house, and try to get as much natural light as possible into it. We used cardboard to build the house. We tested the amount of light it would get into it by turning off the lights in the classroom, and used a lightbulb to resemble the sun. We moved the lightbulb to resemble the sun's movement throughout the day, and recorded the amount of light that all the individual rooms would get at different times of the day.
(# of bulbs) × (Bulb wattage) × (average # of hours per bulb)
(1000 watts/kilowatt )
As a group we had to design a 1,000 square foot house, and try to get as much natural light as possible into it. We used cardboard to build the house. We tested the amount of light it would get into it by turning off the lights in the classroom, and used a lightbulb to resemble the sun. We moved the lightbulb to resemble the sun's movement throughout the day, and recorded the amount of light that all the individual rooms would get at different times of the day.
Site selection:
Even though we weren't going to build another solar house this year, we still went through the process of finding "the perfect spot" for a house. We learned about multiple important conditions/elements that we should keep in mind while choosing a site. My group focused on decent sun exposure, sturdy location/soil, private (but not too secluded), noise levels, and making sure that the site has a nice view. We settled on the site that we found behind the turf field. It isn't too hard to get to, it has a pretty view, and it had a good amount of sun exposure.
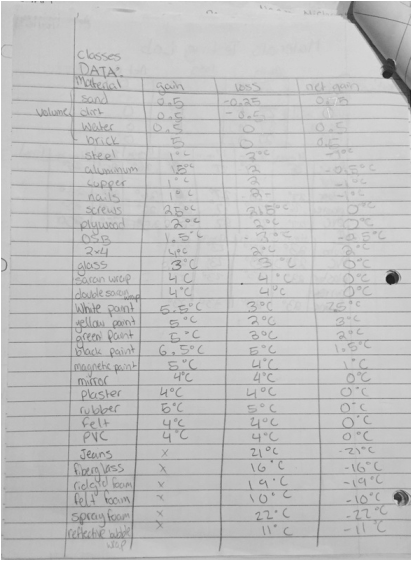
Materials testing lab
As a class we made a materials list for the cold frames and possibly the north window design. Each group got to volunteer to test a certain kind of material. We heated the materials up for 16 minutes with a light bulb, recording temperature changes every 4 minutes. We then turned off the lightbulb and recorded the cooling temperatures every 4 minutes for 16 minutes. Once everyone was done collecting data, we put our numbers that we got onto the board, and analyzed it. We quickly realized that some items were good conductors, while other materials weren't affected by the heat or simply absorbed the heat. My group tested the mirror, plaster, rubber, felt, and PVC. We found out that the rubber conducted the most heat.
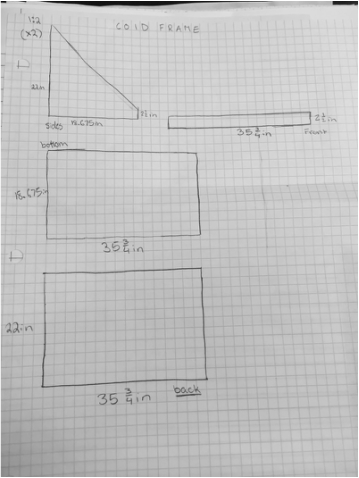
Cold Frame design:
|
This year, instead of building a new solar house again, we decided to design a cold frame and a north window design. The cold frame's job is to act like a greenhouse, letting light in, yet keeping the plants nice and warm. My group decided to create the Hydroframe. We use a hydraulic design to minimize the amount of water that is needed to keep the plants, yet maximize the space inside the cold frame. We painted the inside of the frame maroon, so the color that the plant needs goes to the plant, and the rest is absorb and transformed into heat, keeping the box nice and warm. We also have a special design, where the plants a place in one inch (in diameter) holes, in a clear, hard plastic strip. We would place the seeds into coffee filters, thin enough to let the water reach the plant, but thick enough so it won't break.
North Window design:
|
justification:
As a class we talked about how much energy we humans use. We then researched alternative ways of generating electricity, and the pros and cons of coal, natural gas, hyrdo, solar, and wind energy. Some groups also looked up the economic cost of electricity, the greenhouse effect, and causes of climate change. (The document that we worked on is linked below.)
WInd Turbine:
We learned about hawts and vawts while building our wind turbines. A hawt is a wind turbine with a horizontal axis. A vawt is a wind turbine with a vertical axis. According to our data, the hawt was much more efficient. With the vawt, the blades are being pushed into two different directions at the same time, causing it to move much less efficiently.
Concepts:
Pressure- amount of force in a given area.
Temperature- the measure of how hot or cold something is.
Heat- the amount of thermal energy in an object.
Specific Heat Capacity- a measure of how much energy / heat a substance can hold.
Conduction- transfer of heat through solids
Convection- transfer of heat through fluids
Radiation- transfer of heat through waves/particles/rays
Protons- have a positive charge
Electrons- have a negative charge
Neutrons- are neutral
Evaporation- is the cooling process for water.
Condensation-is the heating process for water.
Vertical light- (90 degrees) is the brightest and most concentrated light, therefore it gives the most light and heat per unit area.
Slanted light- less concentrated and spread out over a larger area, therefore it gives less light and heat per unit area.
Solid- when all molecules are held in place.
Liquid- when all molecules are bonding with each other but they can change bonds to flow (more easily)
Gas- molecules don't have to/can't bond with each other, more movement, and take up way more space.
Archimedes' Principle: an immersed object is buoyed by a force equal to the weight of the fluid it displaces.
Deeper water = more pressure and more density
Ideal Gas Law- (pressure)(volume) = (number of molecules)(constant)(temperature)
Boyle's Law- P1V1=P2V2 (starting conditions = ending conditions)
Bernoulli's Principle- As the speed of a fluid increases, pressure decreases. (low speed=high pressure) (high speed=low pressure)
Zeroth Law of Thermodynamics- explains temperature- if 2 systems are in thermal equilibrium with a third system, ten they are in equilibrium with each other.
First Law of Thermodynamics- Conservation of Energy-energy is neither created nor destroyed; heat is a form of energy.
Second Law of Thermodynamics- entropy increases- disorder increases over time, everything becomes the same temperature.
Third Law of Thermodynamics- Temperature can never get to absolute zero. Heat always exists.
Buoyant floating object- mass of object=mass of water displaced
Temperature- the measure of how hot or cold something is.
Heat- the amount of thermal energy in an object.
Specific Heat Capacity- a measure of how much energy / heat a substance can hold.
Conduction- transfer of heat through solids
Convection- transfer of heat through fluids
Radiation- transfer of heat through waves/particles/rays
Protons- have a positive charge
Electrons- have a negative charge
Neutrons- are neutral
Evaporation- is the cooling process for water.
Condensation-is the heating process for water.
Vertical light- (90 degrees) is the brightest and most concentrated light, therefore it gives the most light and heat per unit area.
Slanted light- less concentrated and spread out over a larger area, therefore it gives less light and heat per unit area.
Solid- when all molecules are held in place.
Liquid- when all molecules are bonding with each other but they can change bonds to flow (more easily)
Gas- molecules don't have to/can't bond with each other, more movement, and take up way more space.
Archimedes' Principle: an immersed object is buoyed by a force equal to the weight of the fluid it displaces.
Deeper water = more pressure and more density
Ideal Gas Law- (pressure)(volume) = (number of molecules)(constant)(temperature)
Boyle's Law- P1V1=P2V2 (starting conditions = ending conditions)
Bernoulli's Principle- As the speed of a fluid increases, pressure decreases. (low speed=high pressure) (high speed=low pressure)
Zeroth Law of Thermodynamics- explains temperature- if 2 systems are in thermal equilibrium with a third system, ten they are in equilibrium with each other.
First Law of Thermodynamics- Conservation of Energy-energy is neither created nor destroyed; heat is a form of energy.
Second Law of Thermodynamics- entropy increases- disorder increases over time, everything becomes the same temperature.
Third Law of Thermodynamics- Temperature can never get to absolute zero. Heat always exists.
Buoyant floating object- mass of object=mass of water displaced
Reflection:
This was a very long project. I think it would have helped to learn all the information needed to accomplish the cold frame and north window project with one group. (So we would have done the solar water heater project and the day lighting design and technique project with one group, and the cold frame and north window project with another group.) A peak in my group were all of the "outside of the box" ideas. Even though it was a peak at times, it also was a pit at others. It was definitely a peak for the cold frame. We followed through with a hydraulic idea that ended up working very well. Since we put so much thought into the cold frame, we lacked with the north window design. We should have communicated better, and made sure that we spent an equal amount of time on both projects. Time management was also one of our pits. We should have managed our time better so we could have put the same amount of time/effort into both of our projects. Other than that, I thought that my group worked well together and had a strong presentation for the cold frame design.